The Joule Experiment
\[ dU = \left(\frac{\partial U}{\partial V}\right)_T dV + \left(\frac{\partial U}{\partial T}\right)_V dT \]
- The second partial derivative is \(C_V\) \[ dU = \left(\frac{\partial U}{\partial V}\right)_T dV + C_V\, dT \]
- What is the first term? Internal pressure \[ \pi_T = \left(\frac{\partial U}{\partial V}\right)_T \]
Measuring the Internal Pressure
- James Prescott Joule (1818-1889) set out to measure the internal pressure
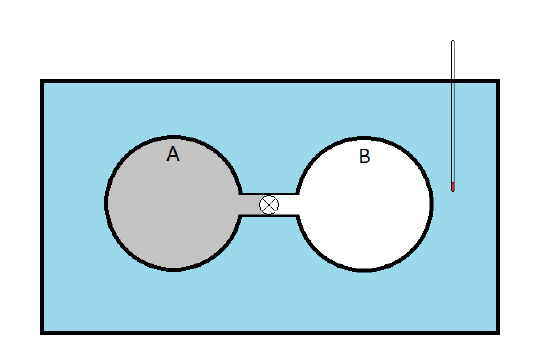
- A has a sample gas while B is evacuated
The Experiment
- When the stopcock is opened, the gas expands into sphere B so \(\Delta V>0\)
- Since B originally had a vacuum, this expansion is done against no external pressure so \(P_{ext}=0\,\therefore\, dw=-P_{ext}\,dV=0\)
- We know that \(dU=dq+dw\) and that \(dU=\pi_T\,dV + C_V\,dT\) so \[ \pi_T\,dV + C_V\, dT = dq + dw = dq \]
- Joule observed no temperature change in the water so he concluded that \(dq=0\) and \(dT=0\)
- Since \(dV>0\) (the gas is expanding), Joule concluded that \(\pi_T=0\)
- In truth, \(dT\ne 0\), but it was too small for Joule to detect it
\[ \pi_T=\left(\frac{\partial U}{\partial V}\right)_T = T\left(\frac{\partial P}{\partial T}\right)_V-P \]
Example 4.2: Internal Pressure of an Ideal Gas
What is the internal pressure, \(\pi_T\), of an ideal gas?
\[ \left(\frac{\partial U}{\partial V}\right)_T = T\left(\frac{\partial P}{\partial T}\right)_V-P \]
\[ PV_m=RT \]
\(\;\)
Example 4.3: Internal Pressure of a van der Waals Gas
What is the internal pressure, \(\pi_T\), of a van der Waals gas?
\[ \left(\frac{\partial U}{\partial V}\right)_T = T\left(\frac{\partial P}{\partial T}\right)_V-P \]
\[ P=\frac{RT}{V_m-b} - \frac{a}{V_m^2} \]
The Joule-Thomson Effect
- In 1852, Joule Began working with William Thomson (who would later become Lord Kelvin)
The Joule-Thomson Experiment
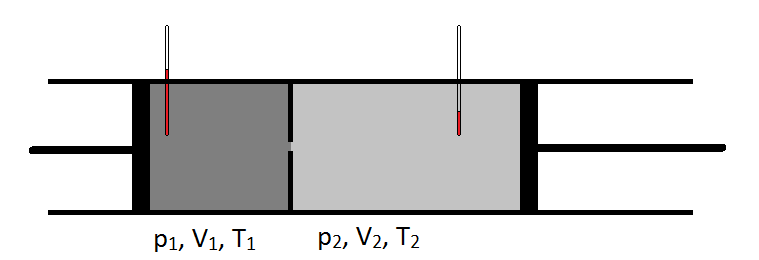
- Gas is pumped into the left side of a lead pipe at a constant rate
- The pipe has a constriction in it so that the pressure in the left chamber is higher than the right chamber.
- The temperature is monitored in each chamber.
In theory, the temperature should increase when the volume increases. Since this experiment allowed volume to increase and pressure to decrease, they were able to observe how temperature is affected when both of these variables are changed.
\(\;\)
The Joule-Thomson Coefficient
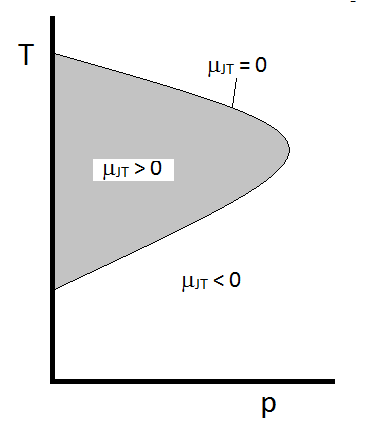
- Not all gases cool as they expand from high pressure to low pressure
- The sign of \(\Delta T\) can be determined by measuring the Joule-Thomson coefficient (\(\mu_{JT}\)) \[ \mu_{JT}\equiv \left(\frac{\partial T}{\partial P}\right)_H \]
Working to Make \(\mu_{JT}\) Measurable
- We can develop an equation for calculating \(\mu_{JT}\) in terms of measurable quantities. But first
\[ dH \equiv \left(\frac{\partial H}{\partial P}\right)_TdP + \left(\frac{\partial H}{\partial T}\right)_PdT \] We will show later that \[ \left(\frac{\partial H}{\partial P}\right)_TdP = -T\left(\frac{\partial V}{\partial T}\right)_P+V \] The second term of which can be written in terms of the isobaric thermal expansivity \[ \left(\frac{\partial H}{\partial P}\right)_TdP = -TV\alpha +V = V\left(1-T\alpha\right) \] \[ dH \equiv \left(\frac{\partial H}{\partial P}\right)_TdP + \left(\frac{\partial H}{\partial T}\right)_PdT \] Divide each side by \(dP\) and constrain to constant \(H\) \[ \left. \frac{dH}{dP}\right|_H = \left(\frac{\partial H}{\partial P}\right)_T\,\left.\frac{dP}{dP}\right|_H + \left(\frac{\partial H}{\partial T}\right)_P\,\left.\frac{dT}{dP}\right|_H \] This can be simplified \[ 0 = \left(\frac{\partial H}{\partial P}\right)_T\,\left(1\right) + \left(\frac{\partial H}{\partial T}\right)_P\,\left(\frac{\partial T}{\partial P}\right)_H \] \[ 0 = V \left(1-T\alpha\right) + \left(\frac{\partial H}{\partial T}\right)_P\,\left(\frac{\partial T}{\partial P}\right)_H \] We can use out definitions of \(C_P=\left(\frac{\partial H}{\partial T}\right)_P\) and \(\mu_{JT}=\left(\frac{\partial T}{\partial P}\right)_H\) \[ 0 = V \left(1-T\alpha\right) + C_P\mu_{JT} \] This can be solved to find that \[ \mu_{JT} = \frac{V\left(T\alpha -1\right)}{C_P} = \frac{V}{C_P}\left(T\alpha-1\right) \]
Useful Definitions and Relationships
- Heat Capacities \[ C_V\equiv \left(\frac{\partial U}{\partial T}\right)_V \;\text{ and }\; C_P\equiv \left(\frac{\partial H}{\partial T}\right)_P \]
- Coefficient of Thermal Expansion \[ \alpha \equiv \frac{1}{V}\left(\frac{\partial V}{\partial T}\right)_P \Rightarrow \left(\frac{\partial V}{\partial T}\right)_P = V\alpha\]
- Isothermal Compressibility \[ \kappa_T \equiv -\frac{1}{V}\left(\frac{\partial V}{\partial P}\right)_T \Rightarrow \left(\frac{\partial V}{\partial P}\right)_T = -V\kappa_T \]
- We have also seen that \[ \begin{eqnarray} \frac{\alpha}{\kappa_T} &=& \left(\frac{\partial P}{\partial T}\right)_V \\ \left(\frac{\partial U}{\partial V}\right)_T &=& T\left(\frac{\partial P}{\partial T}\right)_V-P \\ \left(\frac{\partial H}{\partial P}\right)_T &=& -T\left(\frac{\partial V}{\partial T}\right)_P+V \end{eqnarray} \]
\(\;\)
Example 4.4
Derive an expression for \(\left(\frac{\partial H}{\partial V}\right)_T\) in terms of measurable quantities.
\(\;\)
Example 4.5
Calculate \(\Delta H\) for the isothermal compression of ethanol which will decrease the molar volume by \(0.010\frac{L}{mol}\) at \(300K\). (For ethanol, \(\alpha=1.1\times 10^{-3}K^{-1}\) and \(\kappa_T=7.9\times 10^{-5}atm^{-1}\)).