Radial Distribution Function
The radial distribution function allows us to determine the distance from the nucleus an electron is most likely to be found. To derive this function, we want to consider two components:
- The probability density for a particle with wavefunction \(\psi\)
- The volume of the space a particle (in this case an electron) is allowed to occupy.
Probability Density
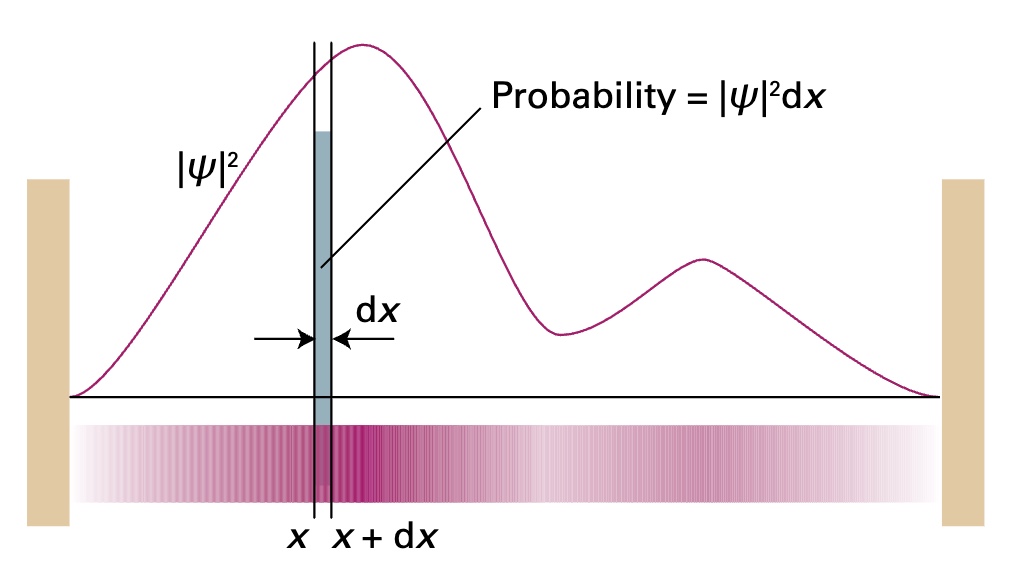
For a fixed radius, the location of a particle is contained in the wavefunction. Max Born1 interpreted the wavefunction in a way that allows us to measure the probability of finding a photon within a region.
Born Interpretation2
If the wavefunction of a particle has the value \(\psi\) at \(x\), then the probability of finding the particle between \(x\) and \(x+dx\) is proportional to \(\vert \psi \vert ^2dx\)
If we vary the radius \(r\), the probability density becomes a function of the radius.
\[probability \ density = \vert \psi (r) \vert ^2\]Volume of a Spherical Shell
For any given distance \(r\) from the nucleus, an electron can be located anywhere in the spherical volume surrounding the nucleus with radius \(r\). To formula for this volume is simply the formula for a volume of a sphere.
\[volume = 4 \pi r^2\]Radial Distribution Function for an s orbital
For a single s orbital, the probability that an electron is found somewhere between the inner and outer surfaces of a spherical shell surrounding the nucleus with radius \(r\) can be found by multiplying the probability density and the volume of the shell. Thus we have
\[P(r) = \vert \psi (r) \vert ^2 \times 4 \pi r^2\]General Form of \(P(r)\)
Consider locating an electron in a generic volume element
\[d \tau = (r^2)dr \cdot (sin \theta) d\theta \ d\phi\]Let the generic wave function for this electron be \(\psi = RY\). Then the probability of finding an electron within this generic volume element can be written as
\[probability \ density = \vert RY \vert ^2 d\tau\]If we want to find the total probability of finding the electron at any angle in a shell of radius \(r\) and thickness \(dr\), we integrate the probability density over the entire surface of the shell. Thus we get
\[P(r)dr = \int_0^{\pi} \int_0^{2\pi} R(r)^2 \vert Y_{l,m_l} \vert ^2 r^2 dr \ sin(\theta) d\theta d\phi\]Since spherical harmonics are normalized to 1, the final generic radial distribution function is
\[P(r) = r^2 R(r)^2\]